
Research Experiences for Undergraduates (REU): Summer 2026 Applications now opened!
Application
Deadline for applications March 2, 2026
For more information contact
Dr. Erica Crespi at: erica.crespi@wsu.edu



10 Weeks
Summer experience (late May – early August) dedicated to learning new skills for success in STEM careers.
$7,000
Stipend provided while you undergo training, plus: benefits, travel and housing expenses, food allowance, and research expenses
10 Positions
Number of positions available for the 2025 summer program: You can be one of them!
Core Values
The foundation of this program is built upon Relationships, Reciprocity, Respect, & Responsibility, plus lots of research skills!
Eligibility
Undergraduate status (freshman-junior when applying) at any college or university with interests in aquatic biosystems and applications to real-world problems
REU students will obtain the following:
- Hands-on research experience using cutting-edge biotechnology
- Access to WSU’s Aquatics Phenomics Research Center, Salmonid Research Center, Outdoor Amphibian Research Facility; may include fieldwork in the Pacific Northwest.
- Field, experimental, bioinformatic, and computational approaches to better understand biological systems
- Deep understanding of relationships, networks, and interactions within biological systems from molecules to landscapes
- Appreciation of how aquatic biosystems interact with human cultural and economic systems
- Scientific literacy and communication skills
- Professional development & networking
- Indigenous knowledge and western science approaches to systems thinking
- Mentoring about career paths that value knowledge of biological systems
Theme 1: Evolutionary biomechanical systems
Evolutionary biomechanics of feeding systems in fishes.
Mentor:
Katherine Corn, School of Biological Sciences
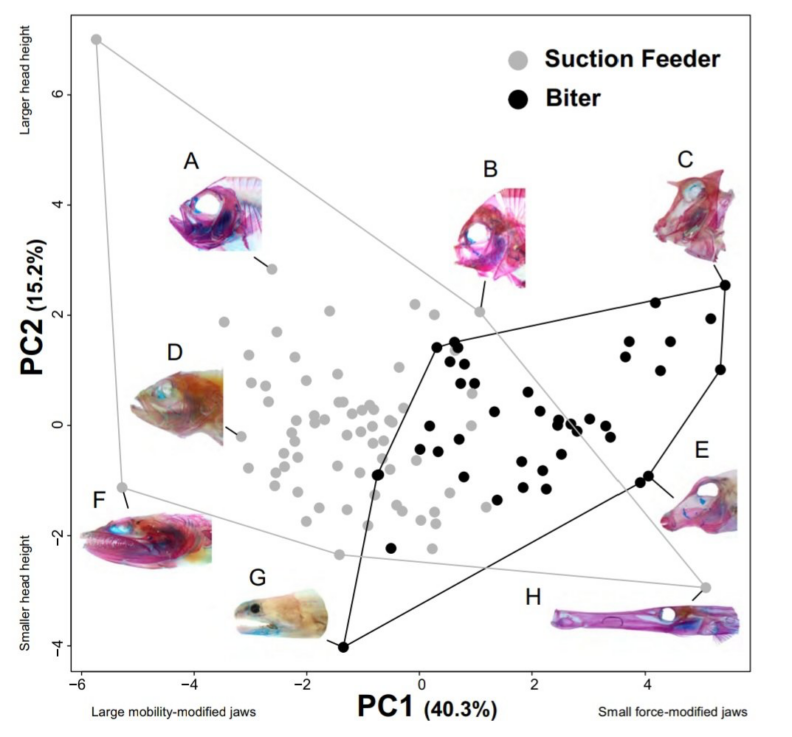
Environmental change can also cause selection for different behaviors, such as feeding, which could drive morphological change and adaptive radiations. Fishes that eat prey attached to the substrate are a key component of aquatic ecosystems. These herbivores and detritivores, collectively referred to as ‘substrate biters’, can be up to 70% of the biomass of aquatic communities; they form a direct trophic link between primary producers and high-level predators; and in coral reef ecosystems, substrate grazers consume algae that competes with coral for precious space. Substrate biters can access a wide range of prey with diverse functional properties, from biting chunks out of sponges to picking at coral polyps or scraping algae from the surface of a rock. How do fishes modify their feeding mechanism in order to better access attached prey, and how does this change with variation in the functional properties of the prey? REU student will use high-speed videography to capture biting strikes by fishes in the laboratory to provide insight into the modifications to the feeding apparatus that are required for capturing functionally diverse prey and shed light on the factors that have contributed to the exceptional radiation of herbivores and detritivores in marine ecosystems.
Theme 2: Integrated systems response to environmental change

Rapidly changing in climates and landscapes are causing dramatic change in aquatic ecosystems such that both animals and plants must be able to tolerate or bounce back from changes in biotic and abiotic factors. Revealing and applying common rules that govern the robustness and resilience of biological systems is an important and indispensable step toward finding solutions for preventing species survival. However, we lack an overarching understanding of the fundamental mechanisms that enable biological systems to appropriately respond to alterations in their environment and withstand or recover from perturbations. Studying biochemical-physiological/sensory-behavioral systems is essential for understanding adaptive responses to environmental change. These researchers span sensory biology, ecotoxicology, cardiovascular physiology and developmental biology to investigate how environmental stressors affect aquatic systems through the interactions of molecular networks, developmental and physiological systems, and organismal-level behaviors and performance. Students interested in integrated systems response to climate change will work in together learning concepts, skills, and research projects:

Intertidal invertebrate systems.
Mentor:
Wes Dowd, School of Biological Sciences
The Dowd lab studies how marine invertebrates not only tolerate, but thrive, in extreme intertidal environments where abiotic conditions vary dramatically over two cycles a day. One line of research in the Dowd lab examines how marine mussels deal with environmental fluctuations that come with living in the intertidal zone. Daily low tides leave these sessile intertidal invertebrates exposed to extreme heat. Furthermore, neighboring individual mussels can reach very different peak body temperatures during the same low-tide period. As a result, they experience unique levels of thermal stress. REU students will be able to study the effects of variable levels of heat stress on individual California mussels, Mytilus californianus. The objective of this project is to assess the effect of heterogenous acclimation temperatures on variation in survival, growth, byssal thread production, and protein expression response of M. californianus after exposure to acute heat stress events. This project includes working directly with the animals in a controlled aquarium setting to simulate tides and extreme environmental events, performing performance analyses and collecting tissue biopsies for physiological measurements, and possibly analyzing large proteomics datasets. In a second avenue of research, REUs will be able to study the physiological consequences of multi-factorial environmental variation in splashpool copepods (Tigriopus californicus). This tiny arthropod is an excellent model system and exhibits remarkable physiological tolerances; salinity, temperature, oxygen, and pH all cycle in sometimes dramatic – and unpredictable – fashion in these splashpools. Performance, including time to first reproduction, whole-animal metabolic rates (in the Loligo respirometry systems), and preferences for different combinations of environmental variables, will be measured on individuals reared under realistic patterns of variation in the laboratory. We will explore the emergent properties of performance under co-occurring variation in salinity, temperature, oxygen and carbon dioxide levels within their current ranges observed in tide pools and in future conditions, which will ultimately better resolve resiliency to environmental change.

Integrated response of multiple stressors in salmonid fishes.
Ryan Shartau, School of Biological Sciences
Animals must respond to environmental change to survive. The Shartau lab studies how fishes tolerate stress in response to natural and human-driven environmental changes. Salmon and other fishes native to Washington State and elsewhere are increasingly stressed by temperature change, hypoxia, and other factors affecting water quality. The objective of this project is to quantify responses to different types of environmental change in fish, including the impact of co-occurring stressors. Students will use state-of-the-art equipment to measure oxygen consumption, behavior, and swim performance under various environmental conditions. The goal of this work is to use physiology to address conservation related questions.
Ecotox-physio-developmental responses to road run-off
Erica Crespi, School of Biological Sciences

Road run-off is affecting water quality in an increasing number of wetlands as impervious surface expands along with urban and suburban areas with increased traffic. A major effect is salinization of wetlands and even ground water due to de-icing salt run-off; in addition, there is the potential for heavy metal run-off, as well as a newly described toxin, 6-PPD-quinone, a leachate from tires, that has been shown to adversely affect some salmonids in the Pacific Northwest. Preliminary work conducted by 2025 REU Nina Allen showed that 6PPD-quinone has similar adverse effects on frog embryos, with developmental delays and vasculature defects similar to those observed in fishes (see inset pictures of Xenopus tropicalis embryos in 6PPD-quinone dose-response experiment: top, control; middle, mid dose, Bottom, high dose). Yet much is unknown about the effects of 6PPD-quinone on other aquatic animals or other developmental stages of amphibians, or how it interacts with salinity to affect development, physiology and overall health of aquatic animals. Furthermore, virtually nothing is known about bioaccumulation or retention times across trophic levels of 6PPD-quinone throughout aquatic communities. REUs interested in filling these knowledge gaps will work in the Crespi Lab and in collaboration with University of Idaho researchers who have derived methods to measure this compound in tissues.
Theme 3: Integrative Host-parasite systems

Disease physiology/immunology and ecology are fields fundamentally built around systems thinking. The study of host-parasite systems spans levels of biological organization, and epidemiological models seek to link how interactions between pathogens and hosts influence the spread of pathogens among host and even populations or communities. Thus, studies are thus ideal for studying robustness and resilience in biological systems. It is imperative we understand the complex ways in human-induced changes in the environment will alter host- parasite dynamics to ultimately be able to predict how anthropogenic change will affect the likelihood and outcome of epidemics. This interdisciplinary research team aims to understand how environmental stressors alter host physiology to alter the severity of disease outcomes at the individual- and population-levels and across life history stages in amphibians. This research has important applications for amphibian conservation and the surveillance of emerging pathogens through the international pet trade. Students interested in amphibian host-parasite systems will work in together learning concepts, building skills, and developing research projects.
Amphibian host-pathogen systems:
Erica Crespi, Jesse Brunner, School of Biological Sciences
Ranavirus – amphibian system under stress
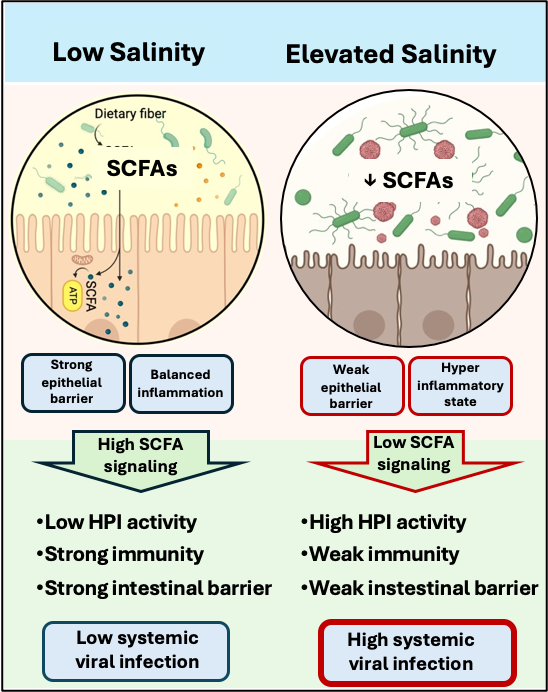
Recent research has shown that chronically elevated salinity experienced in the field or in the lab causes more severe infections of ranavirus, a common water-borne virus, in wood frog tadpoles by increasing stress hormones and reducing immune activity. However, it is unclear the mechanisms that cause this individual-level effect of salinity on host susceptibility or how salinity would affect the likelihood of a widespread epidemic or a mass-mortality event—both population-level processes. Potential REU projects include several types to explore within-host systems of susceptibility, including gut microbiome-host immune and endocrine interactions that lead to increased disease susceptibility, as well as testing candidate microbial metabolites that enhance immune response as new therapies to fight infections. Potential population-level experiments will aim to parameterize epidemiological models to predict outcomes of infection in amphibians. For example, REUs could use video monitoring systems to observe the number of contacts between larval amphibians in with pathogen exposures to parameterize variance in transmission rates depending on environmental conditions in epidemiological models.

Bd and Bsal – amphibian systems
Another deadly amphibian pathogen associated with population decline is the chytrid fungus, Batrachochytrium dendrobatidis (Bd), and B. salamandrivorans is a closely related pathogen threatening salamander species. Long-term studies of the effects of Bd on Cascades frogs has shown that the juvenile stage right after metamorphosis is when this species is most vulnerable to infection, and that larval environmental conditions influence infection after metamorphosis. REUs could participate in experiments in which environmental conditions vary during larval development and after metamorphosis to determine the mechanisms underlying the effects of larval environment on Bd or Bsal infections, and these findings can inform epidemiological models to try and predict population-level outcomes. As in the ranavirus system, REUs could also test potential new therapies to boost gut microbiome-immune system interactions to fight infections, and test these therapies in semi-naturalistic “mesocosms” (experimental ponds).
Fish host-pathogen systems
Tom Waltzek, College of Veterinary Medicine, Washington Disease Diagnostic Laboratory

Dr. Waltzek’s research team is funded by Oregon Sea Grant, USDA, and the CDC to study emerging infectious diseases threating aquaculture, wildlife, and public health. Their research focuses on the evolution, pathology, and disease dynmics of Herpesviruses and Iridoviruses in a variety of fishes, including salmon and sturgeon. He is the Editor in Chief for Diseases of Aquatic Organisms, Assistant Editor for the Journal of Aquatic Animal Health and the Journal of Wildlife Disease, Member of the World Organization of Aquatic Health Committee on Iridoviruses, and Chair of the International Committee on Taxonomy of Herpesviruses and Iridoviruses. REUs interested in aquatic animal health sciences, as well as generally host-pathogen systems, will be working on research aimed at protecting salmon and sturgeon fisheries in an ever-changing climatic landscape.
Application and Selection Procedure:
Students will apply to the NSF ETAP website. Applications will be accepted between Dec. 10, 2025 -Feb 16, 2026. The program will be open to first through third-year students currently enrolled in 2- and 4-year institutions. Applicants must not have participated in another NSF REU program and must be a US Citizen, US National or US Permanent Resident
The application includes:
- Current institution/major
- STEM courses taken
- GPA
- Name and email of one reference (e.g., instructor, mentor, advisor) that we will contact.
Applicants will be asked to submit short statements describing:
- Interests and motivation for applying to the R&R Aquatic Biosystems REU Program
- Experiences working in collaborative teams
- Career goals
- Which R&R research areas they find most interested in pursuing. Applicants will also rank research areas/faculty of most interest.
Successful applicants will be notified via email by March 14th. If selected applicants decline the offer, we will immediately send out offers to those on the wait list.
Washington State University is a public, land-grant research university dedicated to improving lives by serving the public good. R&R REUs will be living in the Northside Dormitory on the Pullman Campus, which is the largest campus in the WSU system. Pullman sits on the Palouse region within the Columbia River Watershed in eastern Washington, with close access to rivers, lakes and mountains for recreation. R&R REUs will interact with the research community of graduate students, postdoctoral researchers, and research faculty, as well as other undergraduate students participating NSF-funded REU programs during shared workshops and the Summer Research Symposium held at the end of the experience. Please see the following links for more information: About WSU , Pullman Campus, NorthSide Residence Hall

Research Facilities
The REUs in this program will have access to state-of-the-art research facilities that will allow them to make the cross-scale connections of functional biological systems. In addition to the research labs of each faculty, REUs will be able to conduct their research projects in the Aquatics Phenomics Research Center, Salmon Research Center, the Airport Gardens Outdoor Amphibian Research Facility, and the following WSU core facilities: Franceschi Microscopy and Imaging Center and the Stable Isotope Core (both within same building complex as APRC) as well as the Genomics Core, Center for Reproductive Biology Gene Editing Core, and the Washington Animal Disease and Diagnostic Laboratory. Students will have access to WSU Libraries and associated databases and data storage on the secure One Drive cloud. Students will also have access to the Kamiak supercomputing cluster.
Communication of Expectations of Behavior and Reporting of Harassment or Disrespectful Behavior
Students and Mentors are expected to abide by the WSU Standards of Conduct for Students and the WSU Policy Prohibiting Discrimination and Harassment, Executive Policy 15. Discrimination and harassment, including sexual harassment, dating violence, domestic violence, stalking, and sexual assault, are prohibited at WSU. Concerns under these policies can be reported to the Center for Community Standards or Compliance and Civil Rights, respectively. Both of these offices have policies and procedures for responding to reports, including formal investigative procedures, resolution protocols, and provision of supportive measures and resource referrals. WSU encourages students to report misconduct and disciplinary sanctions are imposed for violation of university policy. Students will be informed of these expectations and reporting procedures upon arrival at WSU via Percipio on-demand training sessions.
Training Sessions (total time – about 1.5 hours) will include:
- Overview of Executive Policy 15: WSU Policy Prohibiting Discrimination and Harassment
- Compliance and Civil Rights Reporting Options
- Microaggressions
- Title IX: Know Your Rights courses
In addition, Crespi (Program Director), students, and mentors will have an informal discussion session facilitated by someone from the Office of Compliance and Civil Rights during the first week’s activities to review expected behaviors and reporting procedures tailored to the lab, animal facilities, field, and on campus, with reference to these training videos This session will give students the opportunity to ask questions and facilitate trusted relationships with mentors.

2025 Robustness and Resilience of Aquatic Biosystems REU Mentee List
Erica Crespi, Director: erica.crespi@wsu.edu
Grace Curtis, Assistant Director: grace.curtis@wsu.edu
| First and Last Name | Current Enrollment Institution Name | Primary Mentor(s) | Team and Project |
| Ruby Moore | Florida State University | Dr. Katherine Corn | Biomechanical systems of fishes: The Evolution of the Intramandibular Joint in Reef Fishes |
| Sofia Yildirim | Saint Mary’s College of California | Dr. Katherine Corn | Biomechanical systems of fishes: Feeding Performance Across Functional Niche Space |
| Jessica Matthew | University of South Dakota | Dr. Jen Wisecaver | Genomic systems and algal blooms: Interspecific Variation in Growth Rate in the Cryptic Species Complex, Prymnesium parvum |
| Nina Allen | University of Wisconsin Madison | Dr. Zhihua Jiang/Dr. Erica Crespi | Genomic systems of aquatic organisms: Phenomics of 6PPD-quinone Exposure in Xenopus tropicalis Embryos |
| Tammi (Jaralynn) Morellano | University of Tulsa | Dr. Zhihua Jiang | Genomic systems of aquatic organisms: Molecular Response to Hydrostatic Pressure Throughout Zebrafish Embryogenesis |
| Chris Close | Eastern Washington University | Dr. Jesse Brunner | Host-pathogen systems: Testing the EIicacy of Biosecurity Practices in the Ranavirus-Lithobates sylvaticus System |
| Nicholas Kessler | Washington State University | Dr. Jesse Brunner | Host-pathogen systems: Sequential Contact and Disinfection: Testing Aquarium Net Fomite-mediated Transmission of Ranavirus in Wood Frogs |
| Tamia Love | North Carolina A & T State University | Dr. Erica Crespi | Host-pathogen systems: Mitigating Batrachochytrium dendrobatidis Infections in Juvenile Wood Frogs Through Dietary Short-Chain Fatty Acid Supplementation |
| Tyler Robinson | Washington State University | Dr. Erica Crespi | Host-pathogen systems: Butyrate health eIects on Batrachochytrium dendrobatidis infected wood frogs (Lithobates sylvaticus) |
| Ana Rowley | Scripps College | Dr. Wes Dowd | Intertidal invertebrate systems and climate change: Community Physiology in Splashpools: Interactions of Primary Producer and Consumer Responses to Thermal Stress |
| Adel Bordas | Oregon State University | Drs. Georgina Cox and Mike Phelps | Salmonid systems and climate change: Investigating the Role of EGLN3 in Hypoxia Response and Cardiac Physiology in Rainbow Trout (Oncorhynchus mykiss) |
| Jingping Graber | The University of Texas at Austin | Drs. Georgina Cox and Mike Phelps | Salmonid systems and climate change: EIects of Casr Knockout on Cardiac Function, Calcium Regulation, and Bone Mineral Density in Rainbow Trout (Oncorhynchus mykiss) |
| Asher Holloway | Washington State University | Drs. Georgina Cox and Mike Phelps | Salmonid systems and climate change: Examination of the EIects of Egln2 Knockout in Oncorhynchus Mykiss Cardiovascular Function |